
Other compounds are made from different combinations of atoms, like water… carbon dioxide… and table salt.Īnd that’s it! Nearly everything in this room, in your room, and in the entire universe, is made of: elements with one kind of atom, compounds containing different types of atoms chemically bonded together and mixtures of different elements and compounds together. The periodic table captures a subtle pattern that runs through the chemical elements, the fundamental building blocks of everything. Iron sulfide, a new substance, has been formed because the iron and sulfur atoms are now chemically bonded together. First invented by Russian chemist Dimitri Mendeleev in 1869. The iron can no longer be removed by a magnet because it’s bonded to the sulfur. :max_bytes(150000):strip_icc()/accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
Major periodic trends include: electronegativity, ionization energy, electron affinity, atomic radius, melting point, and metallic character. When the iron and sulfur are heated together……atoms of the two elements are now joined together by chemical bonds. Periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of a certain element, including its size and its electronic properties. We can still separate the elements in the mixture. The periodic table organizes elements into groups and periods based on their chemical and physical properties. 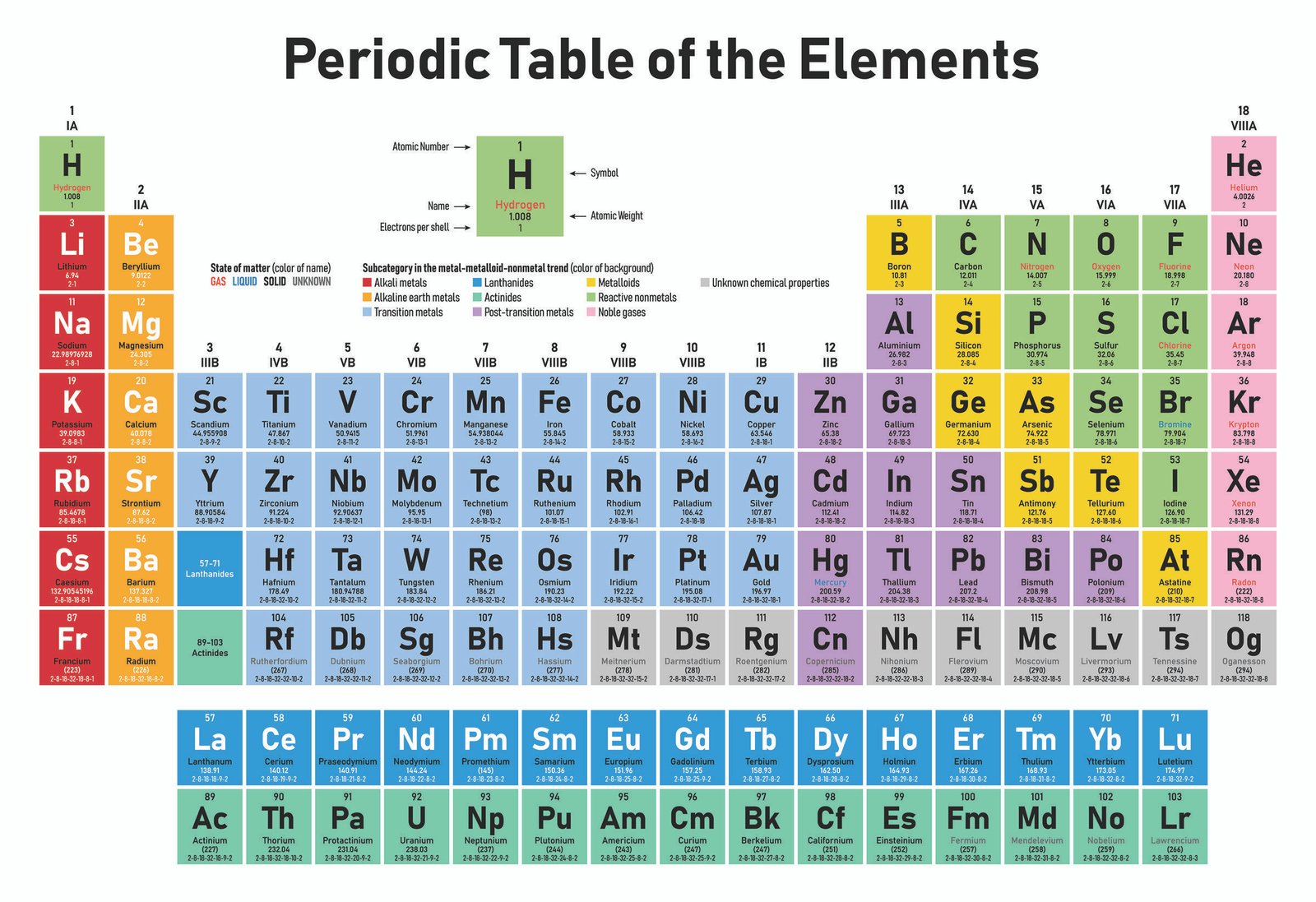
Mass number is often denoted using a capital letter A. In other words, it is the sum of the number of nucleons in an atom. The different elements are not joined together. Mass number is an integer (whole number) equal to the sum of the number of protons and neutrons of an atomic nucleus. This is now a mixture of the elements iron and sulfur. When we mix two different pure substances together, like this, it’s a mixture. The atomic radius of an element tends to increase the further down you go in an element group. It contains only sulfur atoms, and nothing else, so it is pure. No matter what criteria you use to describe the atomic radius, the size of an atom is dependent on how far out its electrons extend. Chemistry - Mendeleev, Periodic Table, Law: Kekule’s innovations were closely connected with a reform movement that gathered steam in the 1850s, seeking to replace the multiplicity of atomic weight systems with Gerhardt’s and Laurent’s proposal. It is made of only one type of atom: iron atoms. 
The calculated atomic radius (pm) is plotted on the y-axis. Atomic number is listed in ascending order on the x-axis. It is the chemical variable that determines the kind of bonds that are formed between atoms. Figure 10.6b: A Plot of periodic variation of atomic radius for the first six rows of the periodic table: the intrinsic sizes of all the elements and clearly show that atomic size varies in a periodic fashion. It is defined as an atom’s ability to attract electrons to itself in a chemical bond. Some substances, like particles of this iron, contain only one kind of atom. Electronegativity (E.N) is the general tendency of an atom in a molecule to draw the shared pair of electrons toward itself. Everything we can see and touch, and quite a lot that we can’t as well, is made of tiny particles called atoms. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
